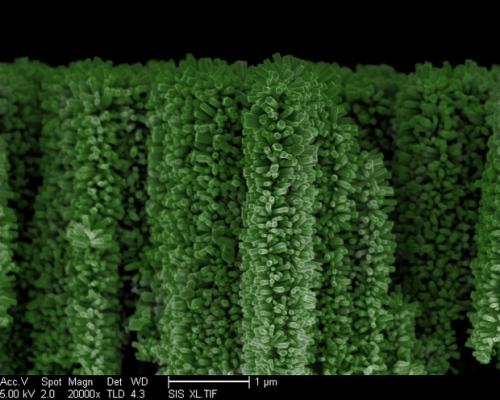
A team of researchers at the University of California, San Diego (UCSD) has developed a method for producing hydrogen using solar energy and nanowire structures that have been nicknamed “nanotrees”. Hydrogen is a fuel that burns cleanly and its only byproduct is water. The problem with hydrogen is the method of production – large amounts of electricity are used to split water molecules into hydrogen and oxygen – electricity which tends to be produced by the burning of fossil fuels. The UCSD team uses a process called photochemical water-splitting to produce hydrogen. The key component of the system is nanowire constructions that look like a forest of trees. Each tree has hundreds of tiny branches, thereby maximizing the surface area of the structure that is able to absorb solar energy. This has been done before, but much less efficiently, using planar solar cells. Research leader Ke Sun explained that the nanotree structures have two distinct advantages over planar systems: the structures allow very small gas bubbles to be extracted faster, thus improving the gas output; and the increased surface area for chemical reactions – which is at least 400,000 times what one would see with a planar system. The research looks promising, but there is one significant limitation. The nanotrees are built from silicon and zinc oxide, both of which are abundantly available. The problem with zinc oxide is that while it is effective in absorbing sunlight, it breaks down over time.It may be that creating the intricate nano-structures would be a time-consuming and expensive process, so long-term usability most likely needs to be a priority. [Image Credit]
A team of researchers at the University of California, San Diego (UCSD) has developed a method for producing hydrogen using solar energy and nanowire structures that have been nicknamed “nanotrees”. Hydrogen is a fuel that burns cleanly and its only byproduct is water. The problem with hydrogen is the method of production – large amounts of electricity are used to split water molecules into hydrogen and oxygen – electricity which tends to be produced by the burning of fossil fuels. The UCSD team uses a process called photochemical water-splitting to produce hydrogen. The key component of the system is nanowire constructions that look like a forest of trees. Each tree has hundreds of tiny branches, thereby maximizing the surface area of the structure that is able to absorb solar energy. This has been done before, but much less efficiently, using planar solar cells. Research leader Ke Sun explained that the nanotree structures have two distinct advantages over planar systems: the structures allow very small gas bubbles to be extracted faster, thus improving the gas output; and the increased surface area for chemical reactions – which is at least 400,000 times what one would see with a planar system. The research looks promising, but there is one significant limitation. The nanotrees are built from silicon and zinc oxide, both of which are abundantly available. The problem with zinc oxide is that while it is effective in absorbing sunlight, it breaks down over time.It may be that creating the intricate nano-structures would be a time-consuming and expensive process, so long-term usability most likely needs to be a priority. [Image Credit] Clean Hydrogen Technology Advances
 A team of researchers at the University of California, San Diego (UCSD) has developed a method for producing hydrogen using solar energy and nanowire structures that have been nicknamed “nanotrees”. Hydrogen is a fuel that burns cleanly and its only byproduct is water. The problem with hydrogen is the method of production – large amounts of electricity are used to split water molecules into hydrogen and oxygen – electricity which tends to be produced by the burning of fossil fuels. The UCSD team uses a process called photochemical water-splitting to produce hydrogen. The key component of the system is nanowire constructions that look like a forest of trees. Each tree has hundreds of tiny branches, thereby maximizing the surface area of the structure that is able to absorb solar energy. This has been done before, but much less efficiently, using planar solar cells. Research leader Ke Sun explained that the nanotree structures have two distinct advantages over planar systems: the structures allow very small gas bubbles to be extracted faster, thus improving the gas output; and the increased surface area for chemical reactions – which is at least 400,000 times what one would see with a planar system. The research looks promising, but there is one significant limitation. The nanotrees are built from silicon and zinc oxide, both of which are abundantly available. The problem with zinc oxide is that while it is effective in absorbing sunlight, it breaks down over time.It may be that creating the intricate nano-structures would be a time-consuming and expensive process, so long-term usability most likely needs to be a priority. [Image Credit]
A team of researchers at the University of California, San Diego (UCSD) has developed a method for producing hydrogen using solar energy and nanowire structures that have been nicknamed “nanotrees”. Hydrogen is a fuel that burns cleanly and its only byproduct is water. The problem with hydrogen is the method of production – large amounts of electricity are used to split water molecules into hydrogen and oxygen – electricity which tends to be produced by the burning of fossil fuels. The UCSD team uses a process called photochemical water-splitting to produce hydrogen. The key component of the system is nanowire constructions that look like a forest of trees. Each tree has hundreds of tiny branches, thereby maximizing the surface area of the structure that is able to absorb solar energy. This has been done before, but much less efficiently, using planar solar cells. Research leader Ke Sun explained that the nanotree structures have two distinct advantages over planar systems: the structures allow very small gas bubbles to be extracted faster, thus improving the gas output; and the increased surface area for chemical reactions – which is at least 400,000 times what one would see with a planar system. The research looks promising, but there is one significant limitation. The nanotrees are built from silicon and zinc oxide, both of which are abundantly available. The problem with zinc oxide is that while it is effective in absorbing sunlight, it breaks down over time.It may be that creating the intricate nano-structures would be a time-consuming and expensive process, so long-term usability most likely needs to be a priority. [Image Credit]